Vaxzevria previously COVID-19 Vaccine AstraZeneca Conditional marketing authorisation granted. Pfizer and BioNTech which developed one of the three COVID-19 vaccines available in the US in May completed their application for full FDA approval for use in.
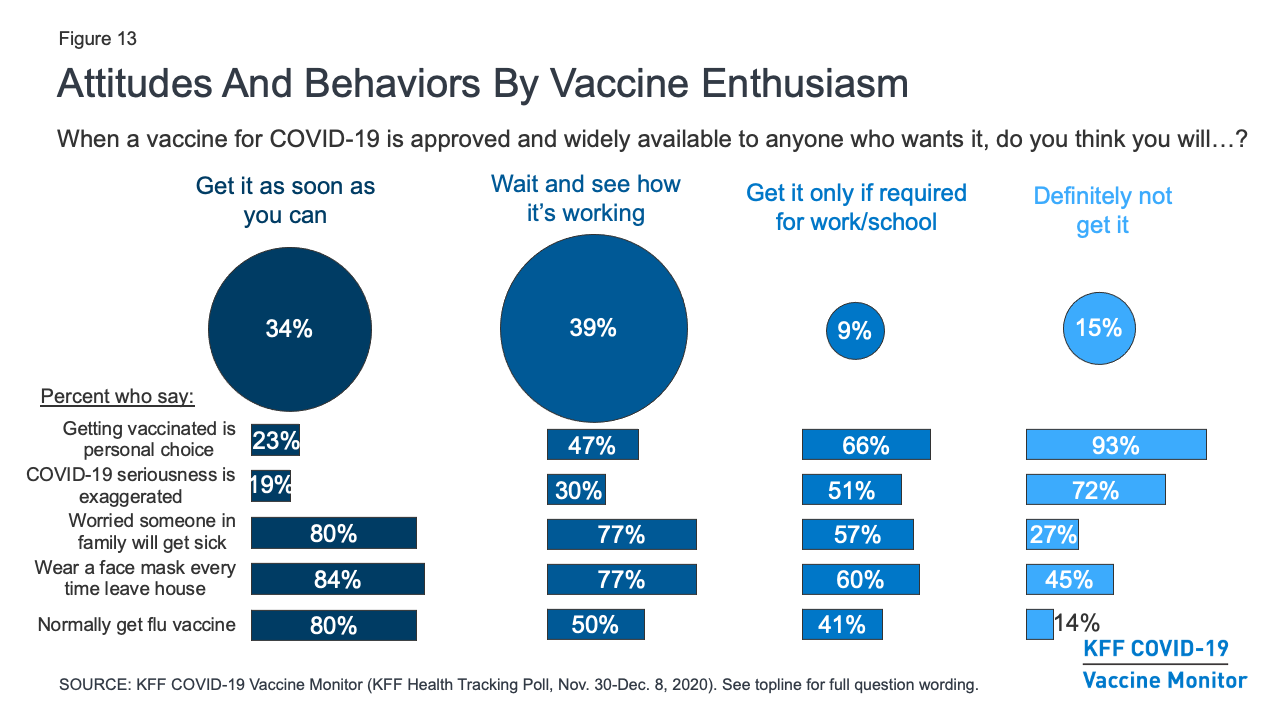
Kff Covid 19 Vaccine Monitor December 2020 Kff
All three FDA-authorized vaccines prevent COVID-19 and serious health outcomes that COVID-19 can cause including hospitalization and deaths.

Fda approved vaccines list covid. Public health experts say they see no reason why the vaccines from Pfizer-BioNTech and Moderna wont receive full approval Johnson Johnson maker of a third vaccine has yet to apply. Janssen Johnson. Two versions of AstraZeneca AZD1222.
Learn how to find a COVID-19 vaccine so you can get it as soon as you can. Its been almost 7 months since the Food and Drug Administration FDA issued the first emergency authorization of a. On Monday the FDAs acting commissioner Dr.
Pfizer BNT162b2COMIRNATY Tozinameran INN Moderna mRNA-1273. Currently no coronavirus vaccine is fully approved. Full FDA approval of the COVID-19 vaccines which are now being given under an emergency use authorization EUA.
Are safe are effective and. SAGE and EUL are complementary but independent processes. The review process could move.
The FDA recognizes that vaccines are key to ending the COVID-19 pandemic and is working as quickly as possible to review applications for full approval an FDA. An FDA slideshow presentation regarding Covid vaccines last year accidentally displayed a long list of possible adverse reactions to the vaccine including myocarditis seizures and even death. The two-dose vaccine which Pfizer.
Its a question many are asking as coronavirus vaccinations continue across the US. The slide showing the FDAs draft list of possible adverse event outcomes appeared briefly during a public meeting by the US Food and Drug Administrations Product Advisory Committee on Oct 22. In fact its one of just seven vaccines that the WHO has approved for inclusion on the EUL.
Food and Drug Administration is aiming to give full approval to Pfizers COVID-19 vaccine by early September according to The New York Times. Sinopharm SARS-CoV-2 Vaccine Vero Cell. All the ones mentioned below have products that have been approved.
EMA recommends COVID-19 Vaccine Moderna for authorisation in the EU. Are the COVID vaccines FDA approved. Experts say full approval may help increase vaccination rates.
Spikevax previously COVID-19 Vaccine Moderna Conditional marketing authorisation granted. Janet Woodcock said the approval should boost public confidence in the. Vaccines are now widely available.
Regulator and to get an approval that puts the vaccine on par with other marketed vaccines. The EUL process is centered on determining if a manufactured product is. These are as follows.
Clinical data login required Paediatric investigation plan. In most cases you do need an appointment. SAGE has issued recommendations on Pfizer 8 January 2021 Moderna 25 January 2021 AstraZeneca 21 April 2021 and Janssen COVID 17 March 2021 vaccines as well as issued a framework for access and population prioritization roadmap.
Do not wait for a specific brand. The Food and Drug Administration is working to approve the Pfizer - BioNTech Covid-19 vaccine on Monday The New York Times reported citing sources. The FDA thoroughly evaluated and analyzed the safety.
The FDA approved seal must represent a high bar -- and premature licensure of a COVID-19 vaccine could seriously damage public confidence in regulatory authorities particularly if long-term. Federal regulators are winding down the process of licensing Pfizers two-dose coronavirus vaccine setting up an approval possibly by Monday and possibly kicking off a wave of new mandates. The vaccine developed by Pfizer and its partner BioNTech is the first COVID-19 vaccine to be subject to a full review by the US.
All currently authorized and recommended COVID-19 vaccines. Reduce your risk of severe illness. 104 Zeilen The FDA expanded the emergency use authorization for the Pfizer-BioNTech COVID-19 Vaccine to include 12 15 year olds and issued an updated FDA COVID-19 Response At-A-Glance Summary.
Sotrovimab Sotrovimab for injection. 9 Zeilen List of authorized drugs vaccines and expanded indications for COVID-19 Authorization holder Brand name proper name or common name Therapeutic area Control number Date of authorization or expanded indication Authorization or expanded indication. Immune sera and immunoglobulins for human use.
The next milestone. The FDA has only granted emergency-use approval of the Pfizer Moderna and Johnson Johnson vaccines but the agency is expected to soon give full approval to Pfizer.

Best Covid Vaccine To Get Comparing J J Pfizer Novavax And Moderna
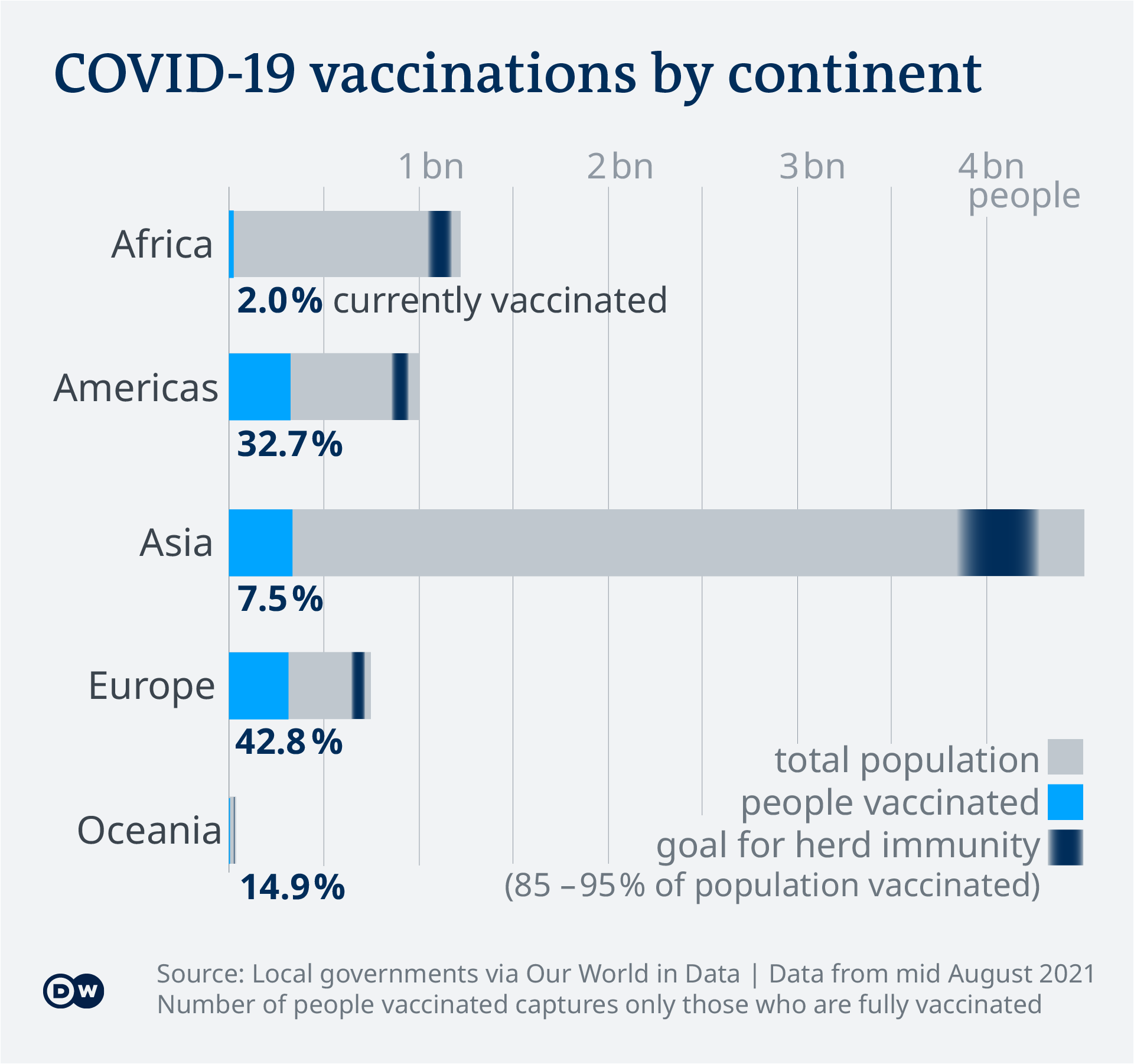
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021

Know Your Vaccines Vaccine Matrix Current Evidence Department Of Health Website

Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021

Fda Advisory Panel Endorses Pfizer Biontech Covid 19 Vaccine
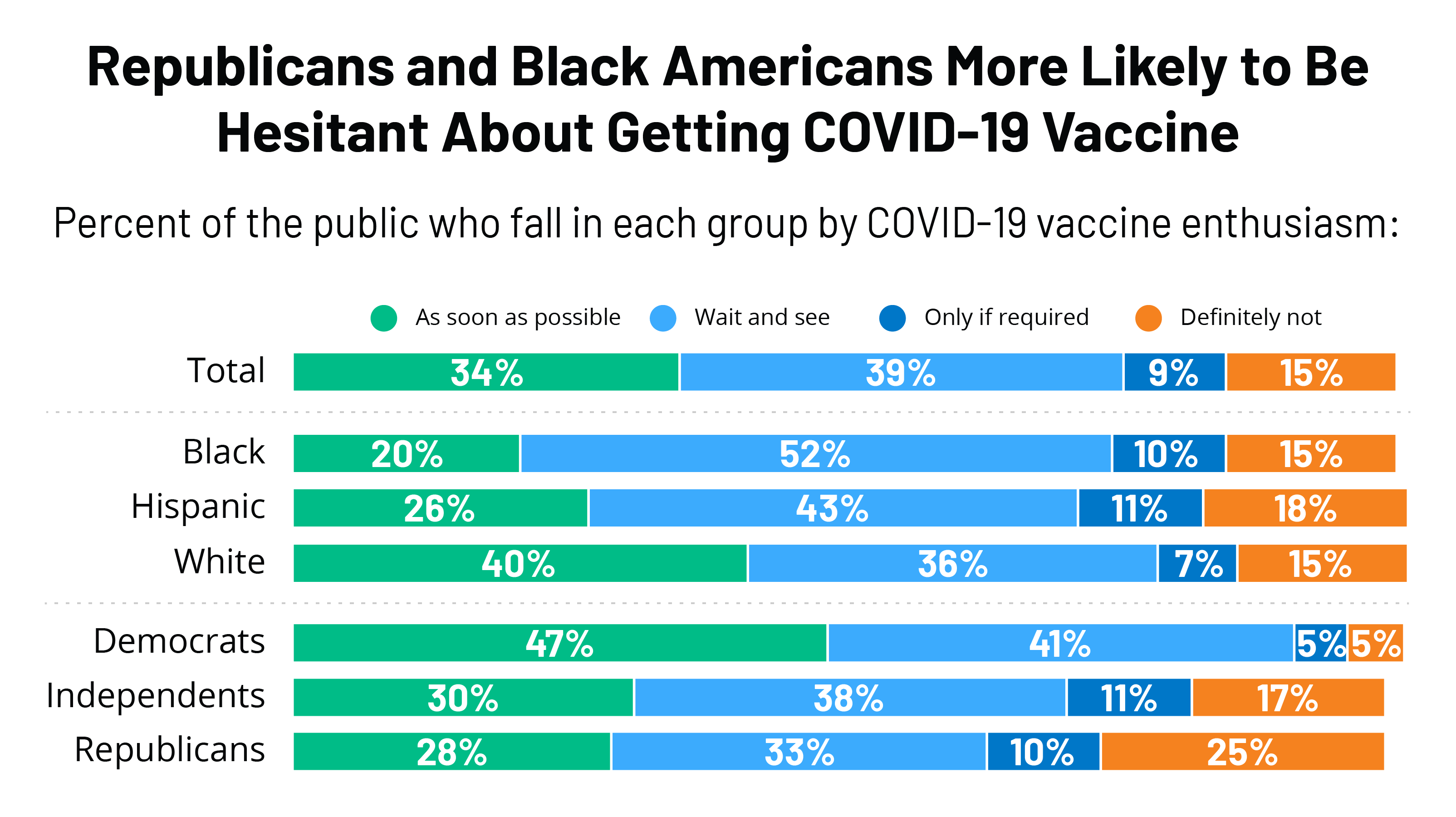
Kff Covid 19 Vaccine Monitor December 2020 Kff
Novavax Announces Further Delays For Regulatory Filings Of Covid 19 Vaccine Pmlive
Covid Vaccines What Full Fda Approval Means For You

Lack Of Fda Covid Vaccine Approval Doesn T Matter Office For Science And Society Mcgill University
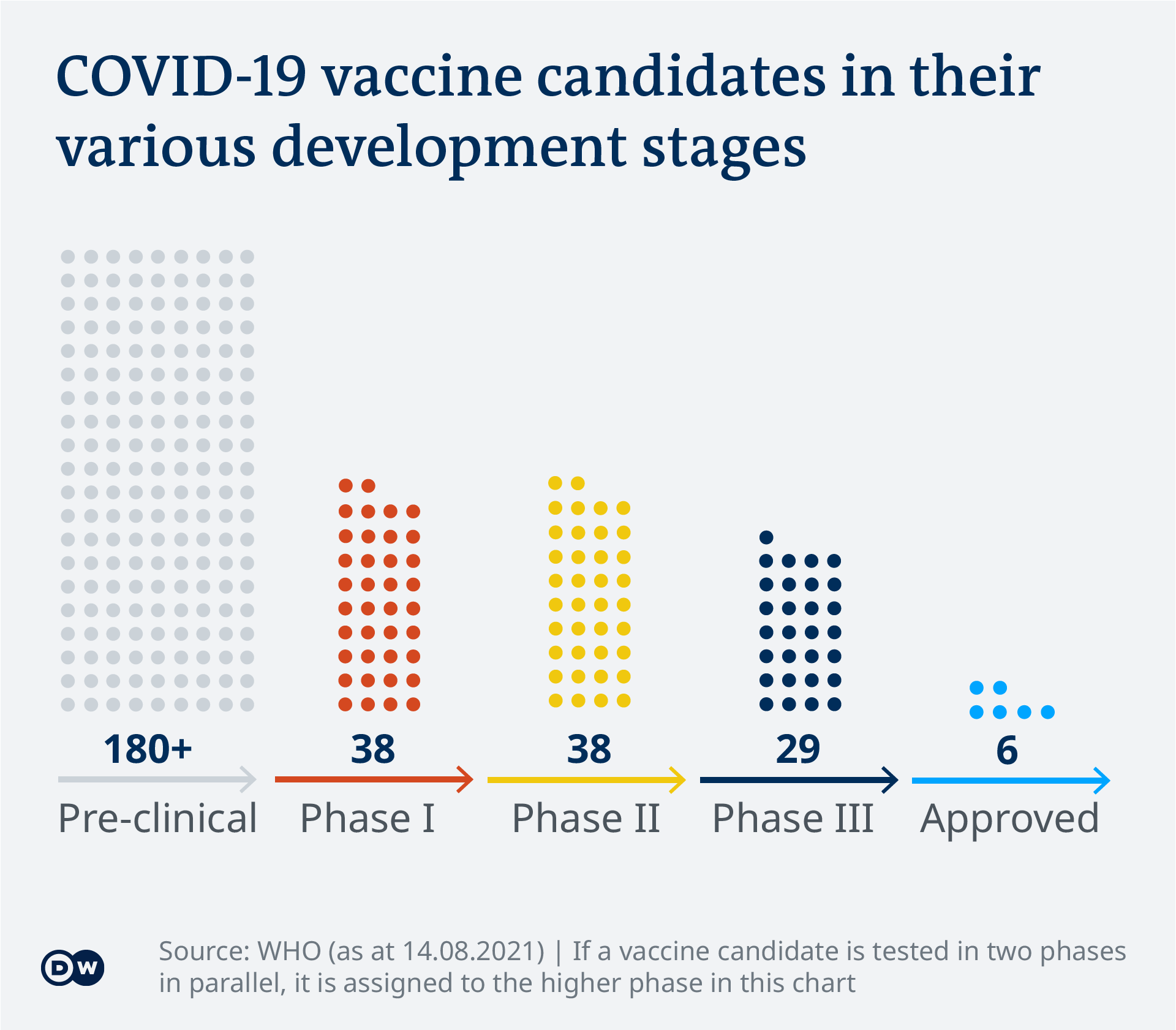
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021
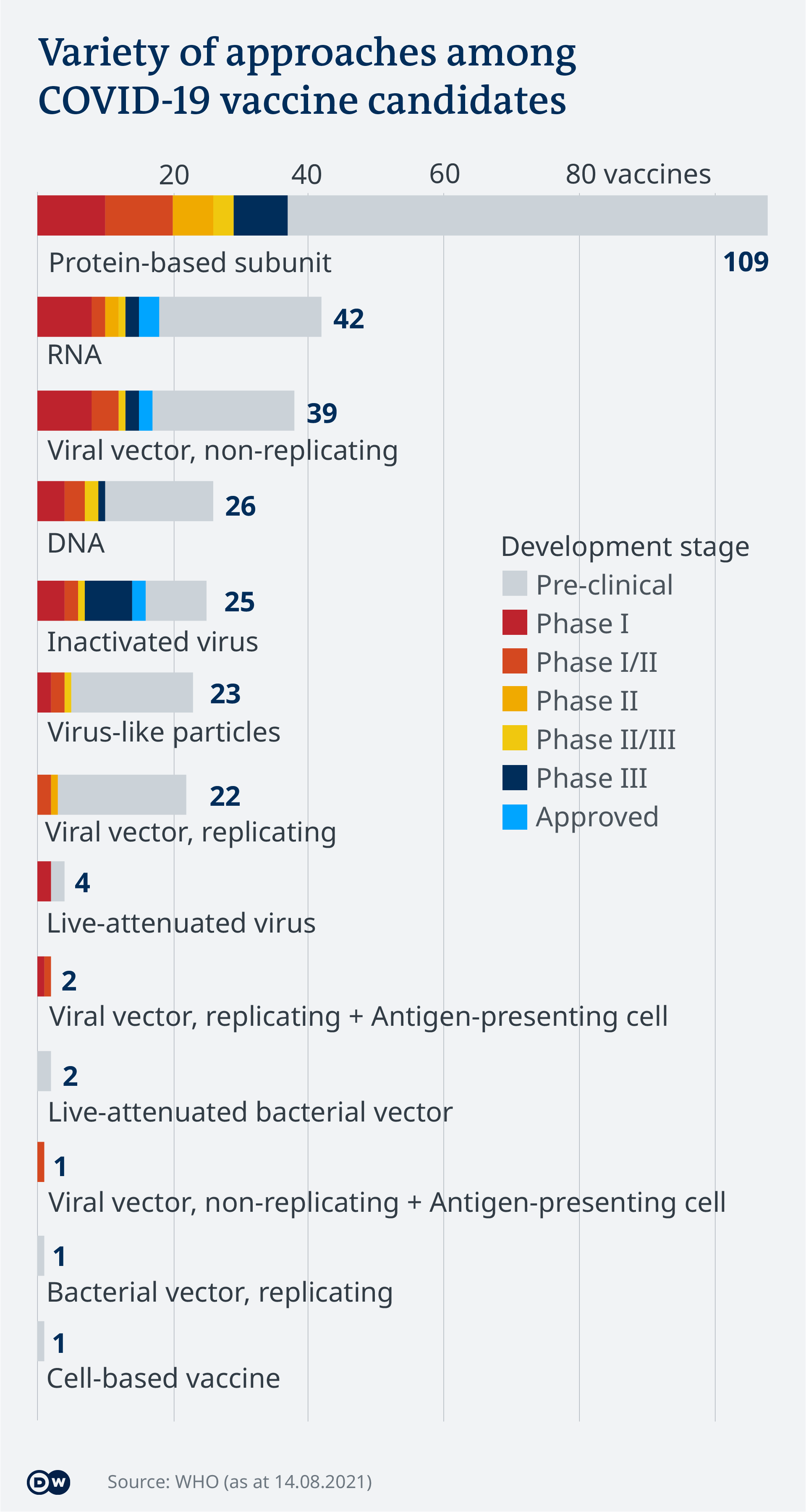
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021
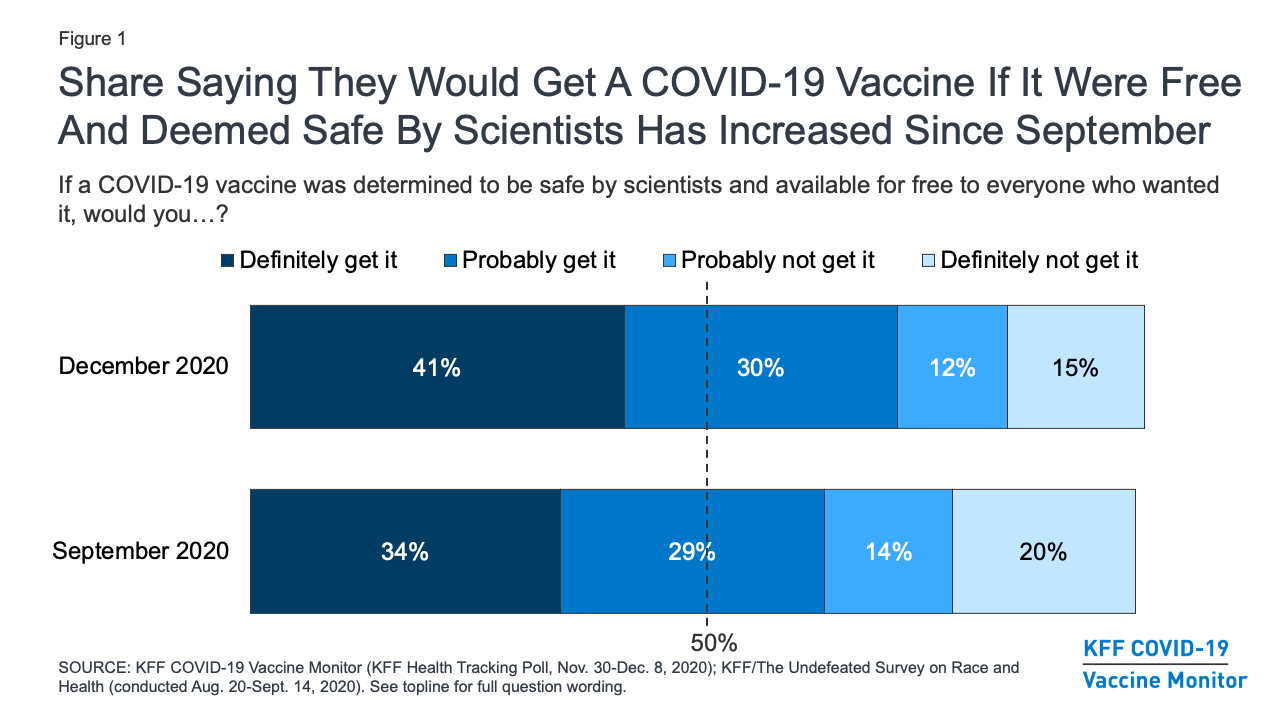
Kff Covid 19 Vaccine Monitor December 2020 Kff
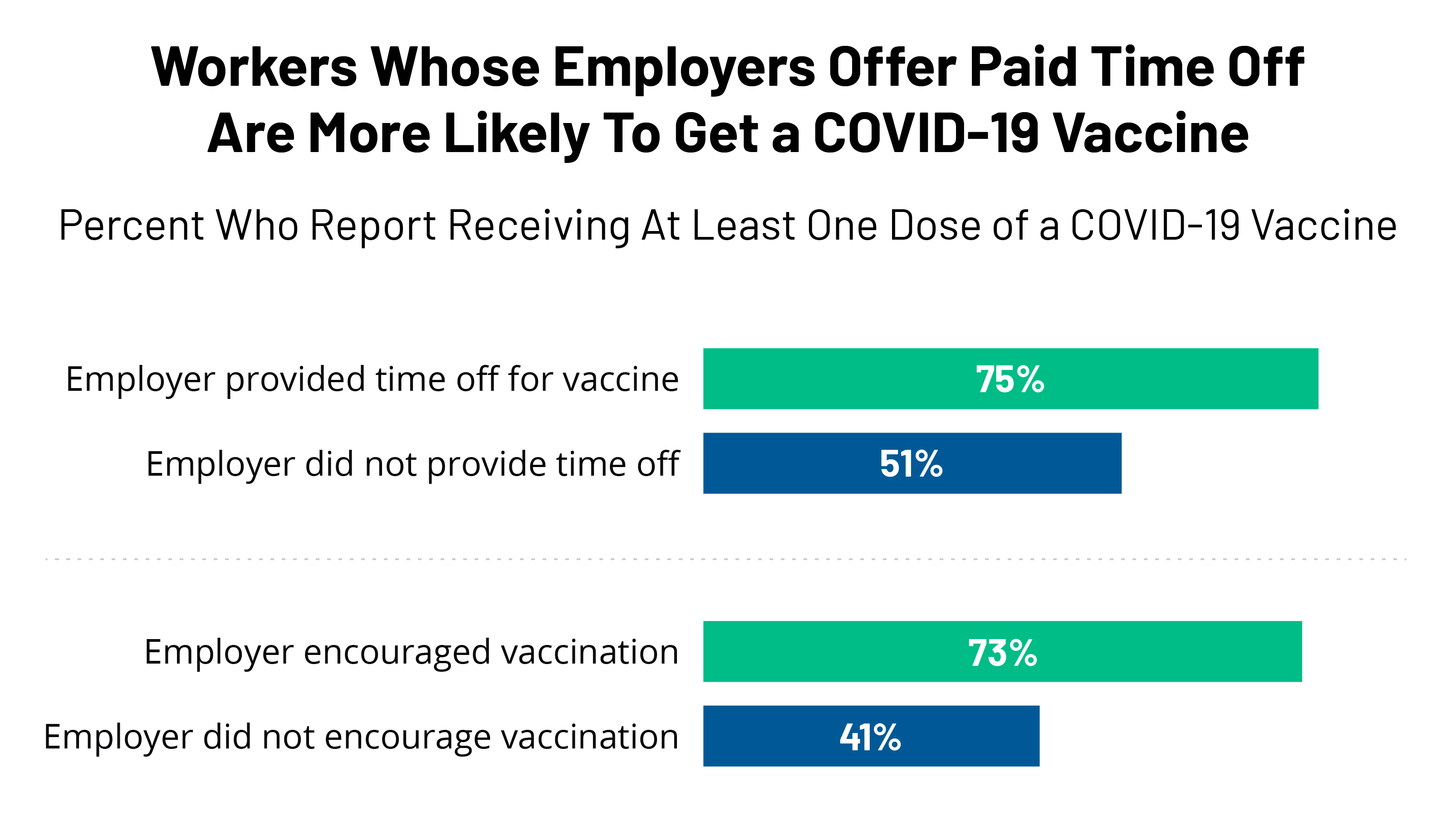
Kff Covid 19 Vaccine Monitor June 2021 Kff
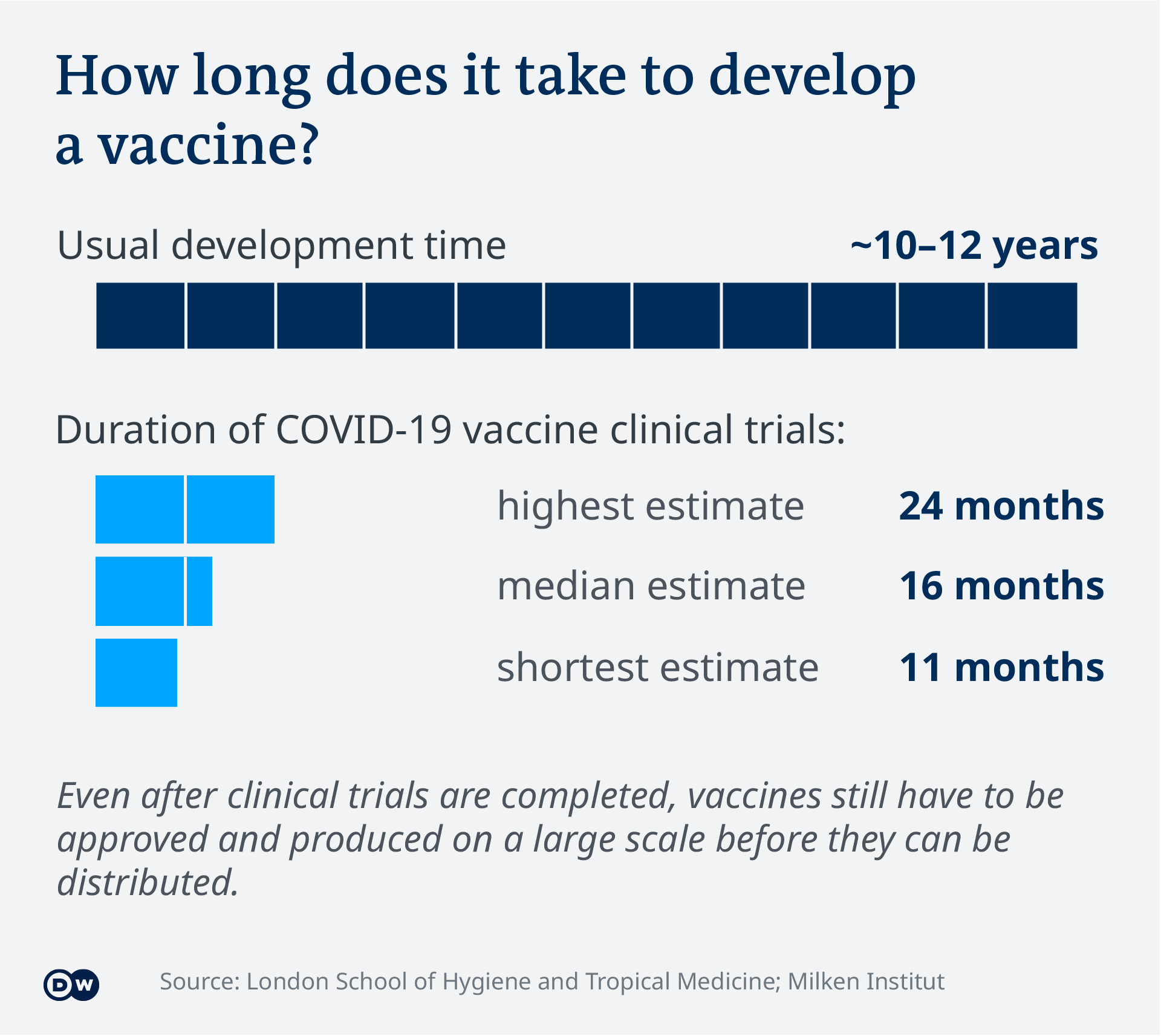
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021

Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021
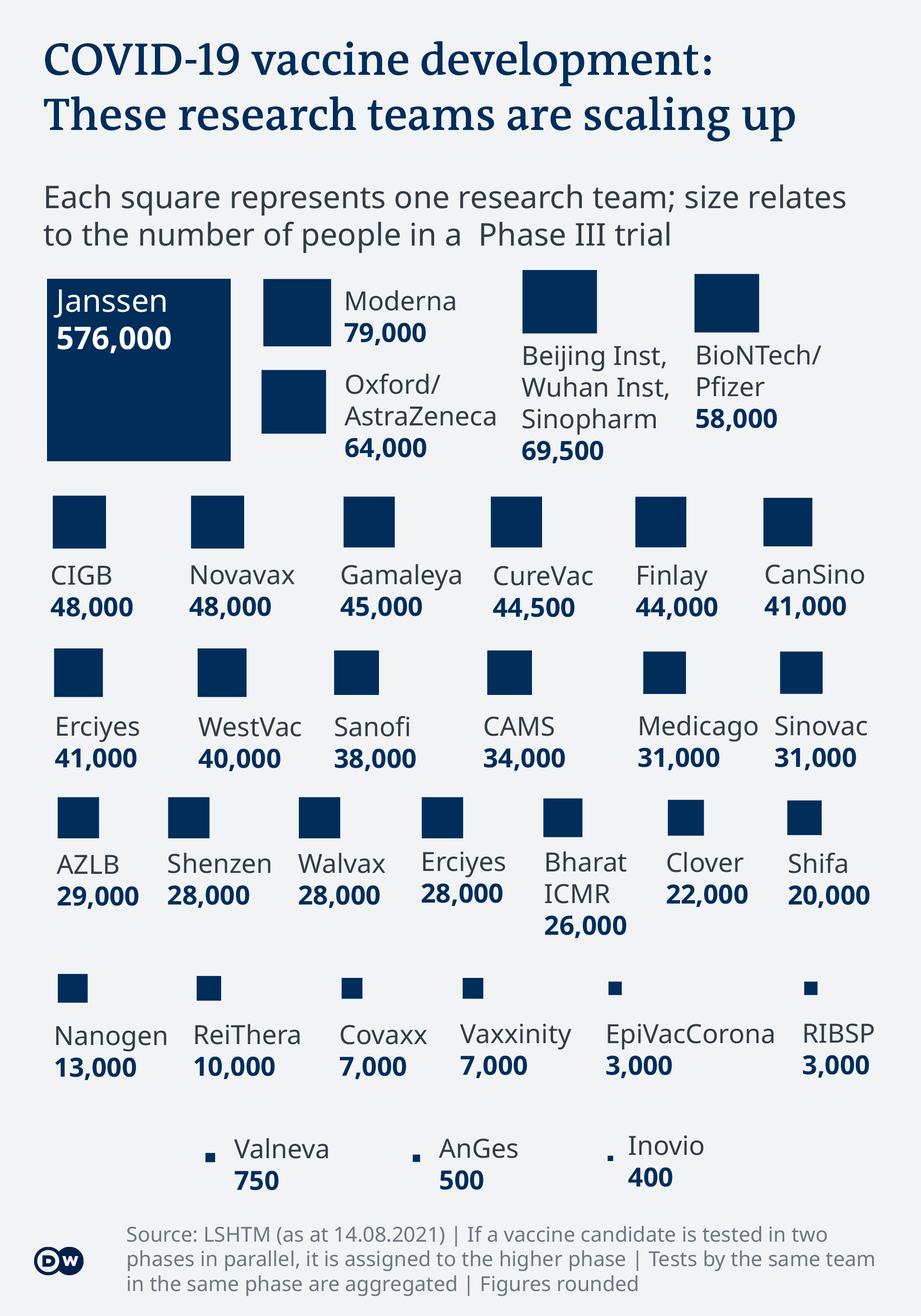
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021
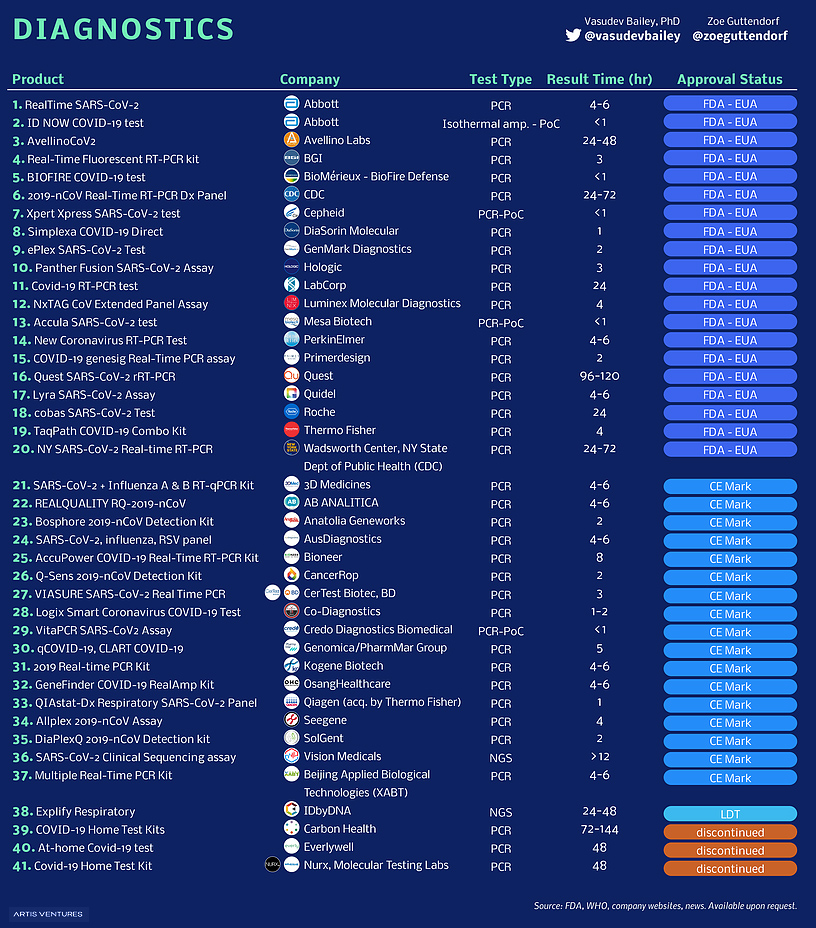
Every Vaccine And Treatment In Development For Covid 19 So Far

Why Two Vaccines Passed The Finishing Line In A Year And Others Didn T And A Month 12 Roundup Absolutely Maybe