Domingo reiterated that at present there is no approved COVID-19 vaccine in the Philippines. Through Executive Order No.
Department Of Health Philippines It Pays To Know The Right Information It Pays To Know The Right Information Read And Know The Right Information On The Covid 19 Vaccines Vaccines Protect
FDA approves emergency use of Sinopharm vaccine.

Fda approved vaccines philippines. At the customs bureau Assistant Customs Commissioner Philip Vincent Maronilla said the agency had received no communication about the arrival of supplies of any COVID-19 vaccine. The Philippines FDA has previously approved Pfizer and BioNTechs coronavirus vaccine. Fda There is as of yet no approved vaccine for the coronavirus disease 2019 COVID-19 in the Philippines Food and Drug Administration Director.
There is no authorized vaccine for Covid here in the Philippines yet that has been given authorization by the FDA He also reiterated that an EUA is not a license to sell the vaccines. All COVID-19 vaccines safe effective once approved by FDA Vergeire Kaya kung ano pong maunang bakuna na dumating atin pong tanggapin yan. Iyong mga ibang country po usually a.
Today the Food and Drug Administration FDA grants its first Emergency Use Authorization EUA to Pfizer-BioNTech COVID19 Vaccine BNT162b2 Suspension for IM Injection Puurs Belgium Site. In the case of the Sinovac vaccine Domingo cautioned. MANILA Food and Drug Administration Director General Eric Domingo on Monday said the Philippines has the second most number of authorized vaccines for use against the coronavirus disease 2019 Covid-19.
2020 vaccines under development. MANILA Philippines Covaxin the COVID-19 vaccine of Indian pharmaceutical firm Bharat Biotech can now be used in the Philippines the Food and. Let us remember the objective of the government is to give us a vaccine to.
MANILA Philippines The decision of the United States Food and Drug Administration FDA to give its full approval to the COVID-19 vaccine developed by Pfizer-BioNTech will hasten the process of allowing its commercial use in the Philippines Malacañang said Tuesday. Pfizer-BioNTech Oxford-AstraZeneca Sinovac CoronaVac Gamaleya Sputnik V Johnson Johnson Janssen Bharat BioTech and Moderna. The FDA Director General by virtue of the Executive Order No.
The Philippines has rolled out AstraZeneca and Sinovac COVID-19 vaccines for its mass immunization which started last March. 104 Zeilen Coronavirus COVID-19 Update. The FDA Chief emphasized.
COVID-19 vaccines from Russias Gamaleya Chinas Sinovac Biotech and. It is for emergency use only and it should be the Department of Health DoH which should procure and administer it. The Philippines Food and Drug Administration FDA has issued an emergency use authorization EUA for Modernas COVID-19 vaccine providing another vaccine option for the Philippines which is.
CHICAGO The US Food and Drug Administration FDA gave full approval to the Pfizer-BioNTechs Covid-19 vaccine on Monday nine months after it rolled off assembly lines under a special emergency use authorization. FDA Philippines Grants Emergency Use Authorization to Pfizer-BioNTech COVID-19 Vaccine. The FDA has found Bharats vaccine is 95 and 92 effective on the study population and among all races respectively.
121 of the President of the Philippines authorizes the issuance of the EUA. The Philippine Food and Drug Administration granted an emergency use authorization for Sinovacs COVID-19 vaccine after a review of late-stage trial. Since last week po we already have seven vaccines that we have given emergency use authorization EUA.
The Philippines Food and Drug Administration has granted EUA to the vaccine candidates of the following firms. Tandaan po natin ang objective po ng gobyerno ay mabigyan tayo ng bakuna para maibsan ang ating sitwasyon So whatever vaccine comes first lets accept that. CNN The US Food and Drug Administration on Monday granted full approval to the PfizerBioNTech COVID-19 vaccine for people age 16 and older.
The Philippine Food and Drug Administration FDA said on Wednesday it has approved the emergency use of the Hayat-Vax COVID-19 vaccine manufactured in. The Philippine FDA has authorized the Pfizer-BioNTech COVID-19 vaccine for emergency use in the country on Jan. The FDA authorized the use under the emergency use authorization EUA for the Janssen COVID-19 vaccine of an additional batch of vaccine drug substance.
Department of Health DOH Undersecretary Maria Rosario Vergeire said that the full approval is also similar to the Certificate of Product Registration CPR of the FDA. Metro Manila CNN Philippines June 7 The countrys Food and Drug Administration on Monday allowed the emergency use of COVID-19 vaccines made. List of COVID-19 Vaccines Authorized by the FDA Pfizer-BioNTech COVID-19 Vaccine BNT162b2 ChAdOx1-S recombinant COVID-19.

Know Your Vaccines Vaccine Matrix Current Evidence Department Of Health Website
Department Of Health Philippines Vaccine Tracker Covid 19 Vaccines Selection And Approval As Of 19 March 2021 Information You Need To Know About The Selection Process And Approval Of The Covid 19 Vaccines
Https Www Fda Gov Ph Wp Content Uploads 2021 03 Eua Sinovac Website 3 1 Pdf

U S Welcomes Arrival To The Philippines Of U S Supported Covax Vaccines U S Embassy In The Philippines

Fda Circular No 2020 036 Guidelines On The Issuance Of Emergency Use Authorization For Drugs And Vaccines For Covid 19 Food And Drug Administration

Fda Advisory No 2021 0664 Public Health Warning On Fake Covid 19 Vaccines Food And Drug Administration
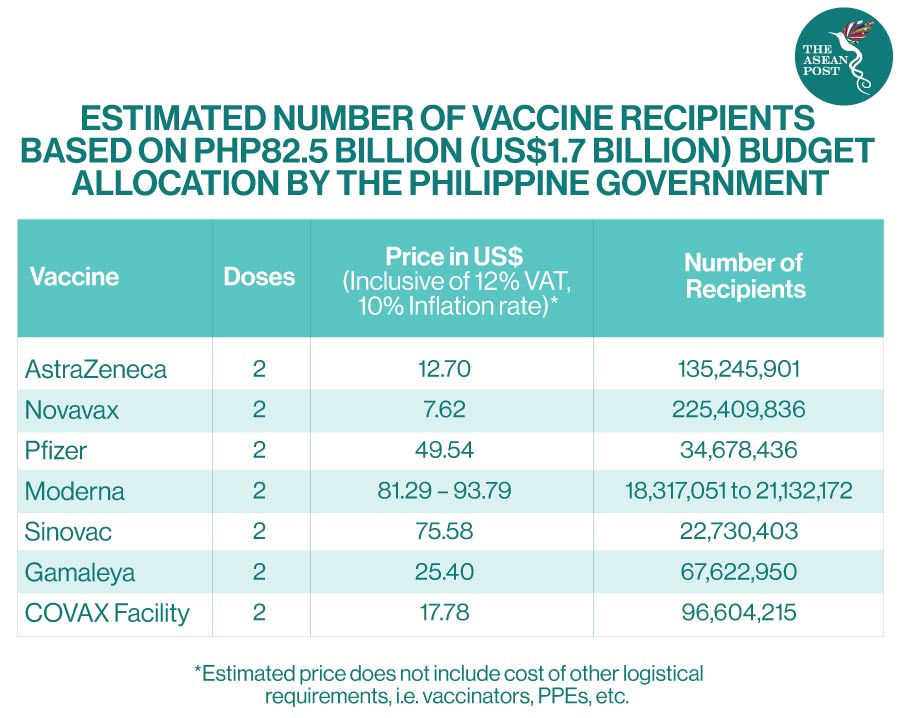
Philippines Roll Out Of Covid 19 Vaccines The Asean Post

Doh Ntf Fda Advisory On Unauthorized Sale Of Unregistered Covid 19 Vaccines February 9 2021 Food And Drug Administration

Know Your Vaccines Vaccine Matrix Current Evidence Department Of Health Website
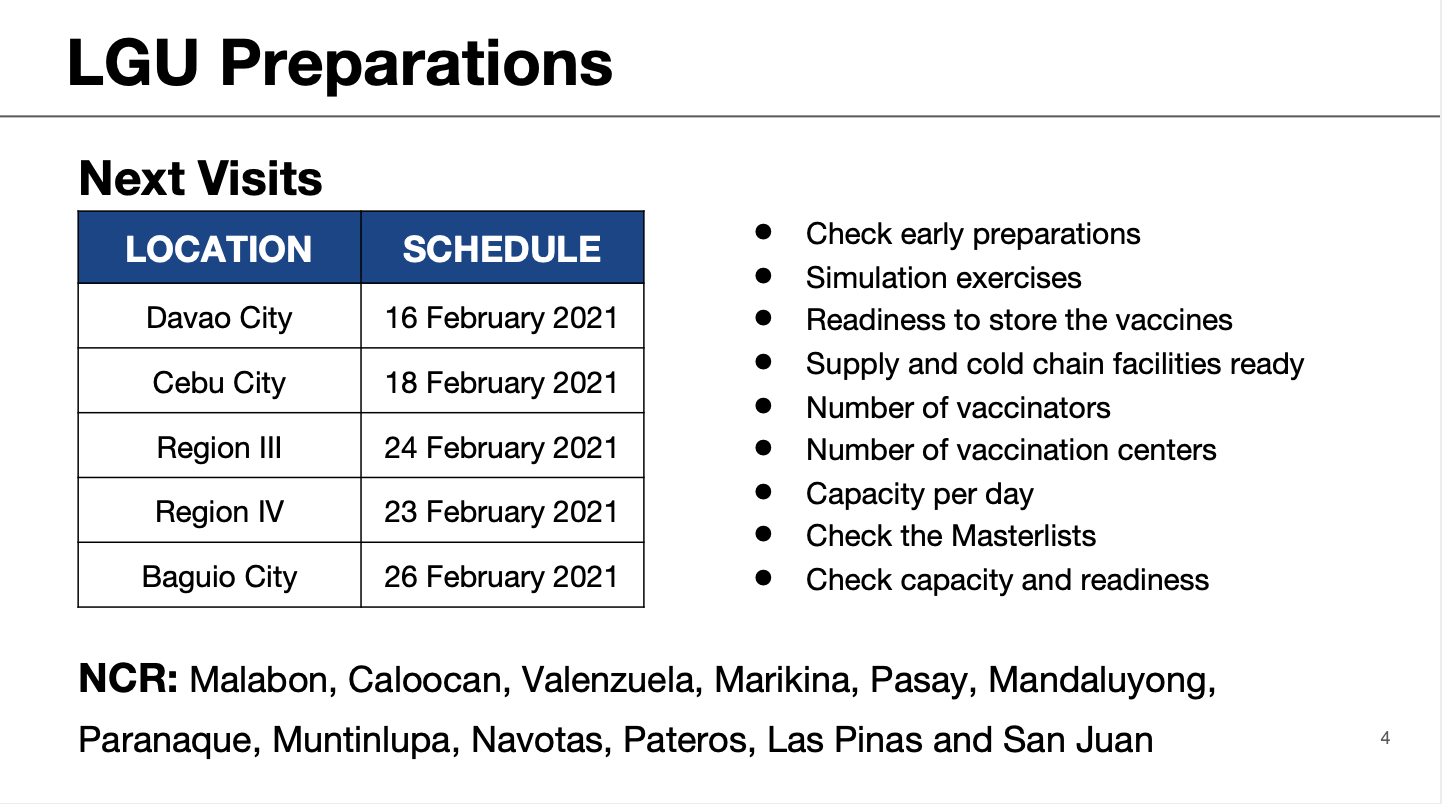
Timeline The Philippines 2021 Covid 19 Vaccine Plan

Timeline The Philippines 2021 Covid 19 Vaccine Plan

Fda Covid 19 Updates Food And Drug Administration

Letter Shows Manila Did Not Seek To Adopt Eua Of Taiwan Vaccine Focus Taiwan

Faqs Emergency Use Authorization Department Of Health Website
Janssen Candidate Vaccine First To Be Approved For Ph Trials Inquirer News

Psmid Philippines Psmidorg Twitter

Department Of Health On Twitter Know Your Covid 19 Vaccines Data As Of March 31 2021 Information You Need To Know About The Covid 19 Vaccines On Our Current Portfolio Technology Platform
Philippines Books 25 Million Doses Of Sinovac S Covid 19 Vaccine Reuters

Fda Philippines Grants Emergency Use Authorization To Pfizer Biontech Covid 19 Vaccine Food And Drug Administration



